Source: The Conversation (Au and NZ) – By Samara Ranie, PhD Student, Institute for Molecular Bioscience, The University of Queensland
The quail is a small, unassuming bird that glides rather than flies and prefers to hide under bushes than to perch on top of a tree. And now, it’s also helping scientists understand serious birth defects in humans.
In a new paper published in Nature Communications, my colleagues and I introduce a new way to study how spinal cords form in the earliest stages of development and what can go wrong – using a genetically modified fluorescent quail embryo.
A fluorescent quail may not be helpful for survival in the wild. But in the lab it allows us to watch living cells organise themselves in real time, revealing processes previously impossible to observe.
What is the neural tube?
The neural tube is the precursor to the brain and spinal cord. In humans, it forms during the first four weeks of pregnancy.
It isn’t much more than a tiny tube stretching from what will become the head to the future tail bone. When this process fails, the neural tube is left open in what is called a neural tube defect.
At least 214,000 pregnancies are impacted by these defects worldwide every year.
In some cases pregnancies are lost; in others the baby will have lifelong disabilities. Spina bifida occurs when the spine is open, most commonly resulting in mobility loss and loss of feeling. Other conditions such as anencephaly occur when this opening is in the brain.
Studying these birth defects is hard because neural tube defects don’t have a single cause. Multiple genetic mutations and environmental factors work in combination.
So how do we even begin to understand what is happening?
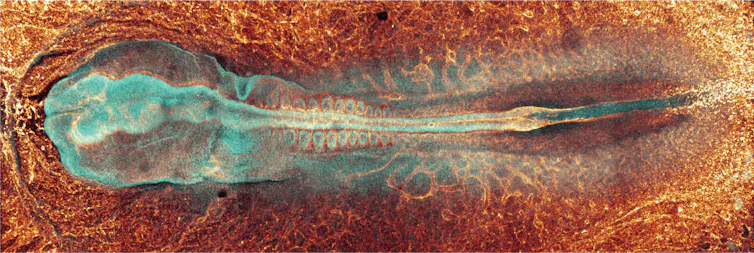
Enter the fluorescent quail
Human spine development is very similar to that of our feathered friends, birds.
Our spines form progressively, one section at a time. Most species only have two sections, but humans and birds – such as quails – develop in three.
The neural tube forms so early in development that studying it in humans with the technology currently available isn’t possible. Even using common animal models, such as mice, is a challenge because we simply can’t see into a uterus to watch this process live and in detail.
Quails, however, grow in an egg. Working out of an egg means that we can study early development with the embryo growing comfortably in its natural environment. Quails also have the benefit of growing quickly, quicker than chickens, so we can study this process as fast as we can.
In 2024 my colleagues created a genetically modified fluorescent quail. With this fluorescent quail we can discover how the neural tube forms during the first 72 hours of development by imaging individual cells to see how they move and send signals to each other as they build the early spine.
But how do we make a quail glow?
A fluorescent tag is added to what we are interested in observing inside the quail cells. That colourful tag can be seen under what’s called a confocal fluorescence microscope. And as the embryo grows, we can watch these tags move around the cell.
Embryos grow for only 24 hours before we cut a window into the egg. The embryo is still smaller than a grain of rice, barely visible on top of the yolk.
But using that fluorescent tag, we record the embryo growing and cells moving and signalling to each other in real time in what I like to consider the “cell superhighway”.
Navigating the cell superhighway
When we video the neural tube, we look close enough to see individual cells moving to their destinations. Cells in this superhighway exist in a state of gridlock that makes the busiest highways in the world look fast. But somehow every cell knows where to go.
Outside the cells, chemical signals draw specific cells to specific locations, like road signs telling you to take the next highway exit.
Each cell also contains an internal “GPS system”, organising left from right, up from down, forward from backwards. Without this signal cells become lost. This is catastrophic for development.
In our new paper, we show that one of these signals – a gene known as PRICKLE1 – plays a crucial role in shaping the early neural tube by coordinating cell movement, but not in the way we previously thought.
PRICKLE1 is crucial for “left versus right” signalling and we know that mutations of PRICKLE1 cause neural tube defects. So by removing PRICKLE1 from the quails we expected “left versus right” signalling to be broken, cells to become lost, and neural tube defects to occur.
But science likes to prove us wrong.
A big discovery from a tiny embryo
We did see that losing PRICKLE1 caused neural tube defects and cells were moving randomly on our cell superhighway. What surprised us was that the “left versus right” signals were completely fine.
Instead, cells could no longer separate up from down.
In our cells almost everything has multiple jobs. There are backup plans and backup-backup plans. But this second, new role for PRICKLE1 seems to not have any backups.
When this up-and-down signalling is damaged, the embryo will develop neural tube defects.
The difference between left and right or up and down seems trivial. But inside the cell, these are distinctly different pathways controlled by different signals.
This discovery will ensure future research can progress in the right direction. It could ultimately lead us to genetic screening methods and therapeutic targets to make a difference to the hundreds of thousands of pregnancies impacted by neural tube defects.
All of this knowledge from our little fluorescent quail embryo, smaller than a grain of rice and less than 72 hours into development.
– ref. Fluorescent quail embryos could help solve serious birth defects in humans – https://theconversation.com/fluorescent-quail-embryos-could-help-solve-serious-birth-defects-in-humans-277090
